Research Projects
Therapeutic and Diagnostic Agents Targeting the Lysophosphatidic Acid Receptor 1
The lysophosphatidic acid receptor 1 (LPAR1) is a G protein-coupled receptor activated by lysophosphatidic acid (LPA), a signaling molecule that functions like a growth factor in various physiological processes, including cell proliferation, migration, and survival. Studies have shown that targeted deletion of LPAR1 in different organ systems leads to physiological effects associated with a range of diseases, such as fibrosis, cancer, and neuropathic pain. Our research focuses on the development of novel molecules targeting LPAR1 through traditional medicinal chemistry approaches, computational drug design, and machine learning techniques. These highly selective molecules can be employed to investigate LPAR1 signaling and its function in relation to cancer progression. Additionally, we conduct applied research to explore the potential of LPAR1 antagonists as therapeutic drugs for cancer treatment and the use of 18F-radiolabeled tracers for cancer imaging with PET.
The lysophosphatidic acid receptor 1 (LPAR1) is a G protein-coupled receptor activated by lysophosphatidic acid (LPA), a signaling molecule that functions like a growth factor in various physiological processes, including cell proliferation, migration, and survival. Studies have shown that targeted deletion of LPAR1 in different organ systems leads to physiological effects associated with a range of diseases, such as fibrosis, cancer, and neuropathic pain. Our research focuses on the development of novel molecules targeting LPAR1 through traditional medicinal chemistry approaches, computational drug design, and machine learning techniques. These highly selective molecules can be employed to investigate LPAR1 signaling and its function in relation to cancer progression. Additionally, we conduct applied research to explore the potential of LPAR1 antagonists as therapeutic drugs for cancer treatment and the use of 18F-radiolabeled tracers for cancer imaging with PET.
Therapeutic and Diagnostic Agents Targeting the Aurora Kinases
The Aurora kinases (A, B, and C) are a family of three isoform serine/threonine kinases that regulate mitosis and meiosis. As master regulators of mitosis, they play essential roles in centrosome maturation, spindle assembly, chromosome alignment, and cytokinesis. Dysregulation, overexpression, or amplification of Aurora kinases have been observed in various cancer types, including lung, breast, colorectal, ovarian, and pancreatic cancers, among others. Specifically, high levels of AURKA and AURKB have been detected in lung cancer cells, correlating with poor prognosis, increased aggressiveness, and resistance to treatment. Our research focuses on developing inhibitors as chemotherapy drugs for the most aggressive lung cancer. Another direction of our research involves creating 18F-radiolabeled tracers targeting Aurora kinases for the early detection of cancer.
The Aurora kinases (A, B, and C) are a family of three isoform serine/threonine kinases that regulate mitosis and meiosis. As master regulators of mitosis, they play essential roles in centrosome maturation, spindle assembly, chromosome alignment, and cytokinesis. Dysregulation, overexpression, or amplification of Aurora kinases have been observed in various cancer types, including lung, breast, colorectal, ovarian, and pancreatic cancers, among others. Specifically, high levels of AURKA and AURKB have been detected in lung cancer cells, correlating with poor prognosis, increased aggressiveness, and resistance to treatment. Our research focuses on developing inhibitors as chemotherapy drugs for the most aggressive lung cancer. Another direction of our research involves creating 18F-radiolabeled tracers targeting Aurora kinases for the early detection of cancer.
What's on our agenda?
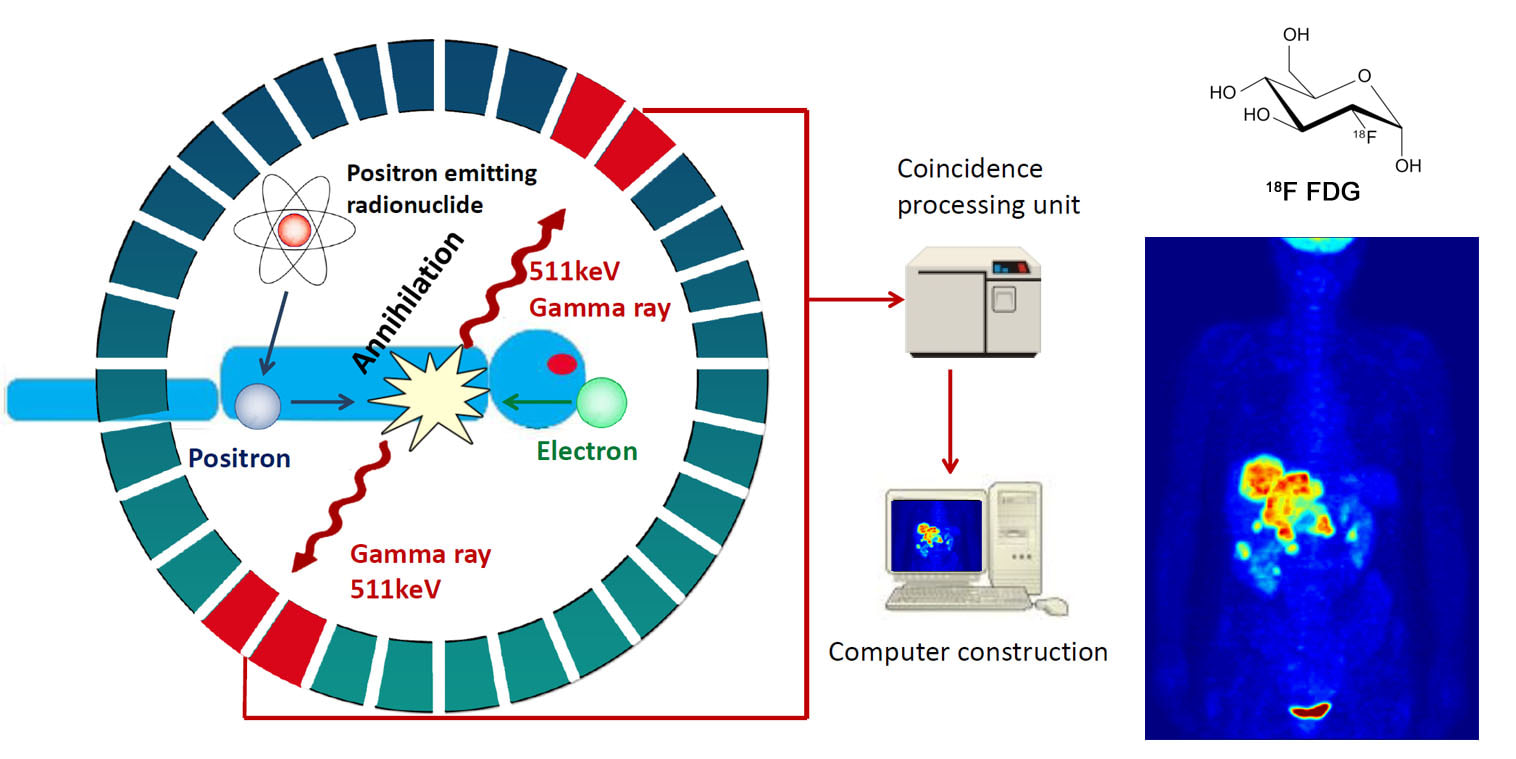
PET Molecular Imaging ModalityPET molecular imaging allows for the observation of biological processes in vivo, and is believed to hold the promise of personalized medicine due to its roles in choosing the right treatment for an individual patient, guiding targeted therapies and developing and optimizing new therapeutics.
Five features of PET: 1) usage of positron emitter, 2) 3D images, 3) non-invasive, 4) high sensitivity, 5) quantifiable amount of radiation. |
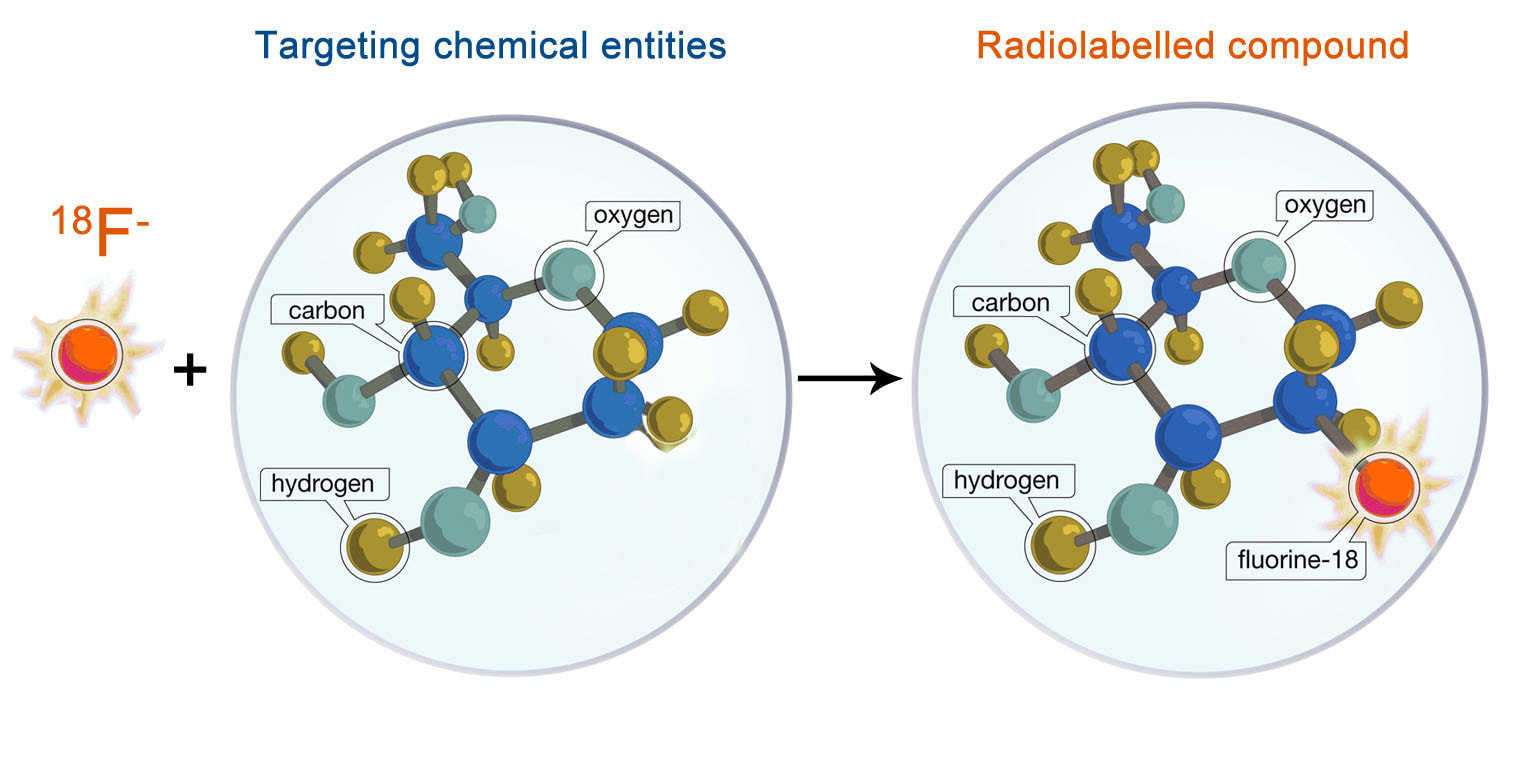
'Hot' ChemistryOrganic chemistry research over the past century has led to significant advances in “cold” fluorination chemistry. However, relative to the vast diversity of synthetic organic chemistry methods, there are still limited radio-fluorination methods currently available. We aim to expand 18F radiochemical space that has previously not or been difficult to access synthetically in 'hot' lab.
|
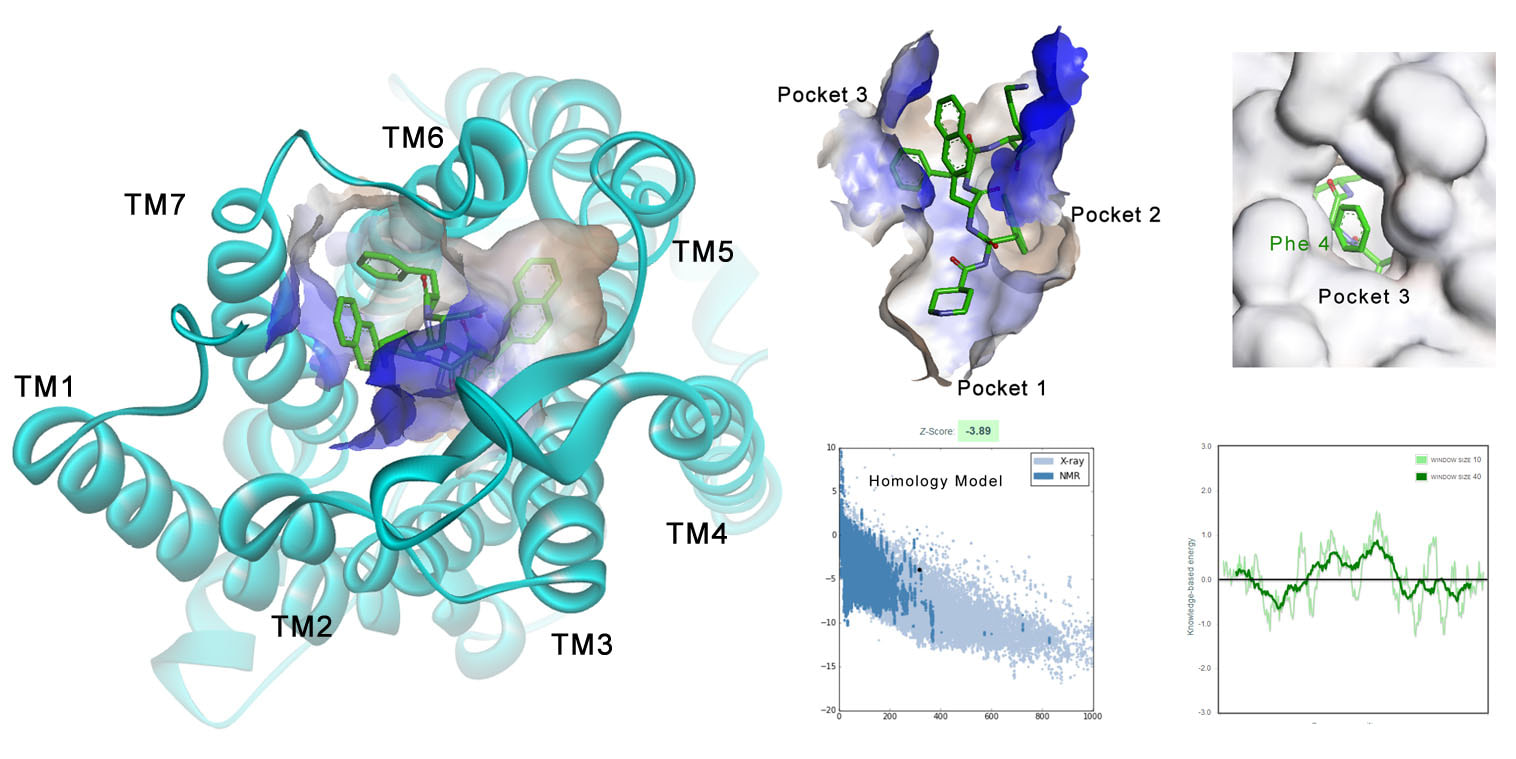
Computational Molecular DesignWe perform ligand-based and structure-based molecular design. Ligand-based method includes 3D-QSAR model, scaffold hopping, etc. Structure-based method includes horology modelling, molecular docking, molecular dynamics simulation. We aim to rationally design drug-like molecules that will have high binding affinity for their protein target.
|
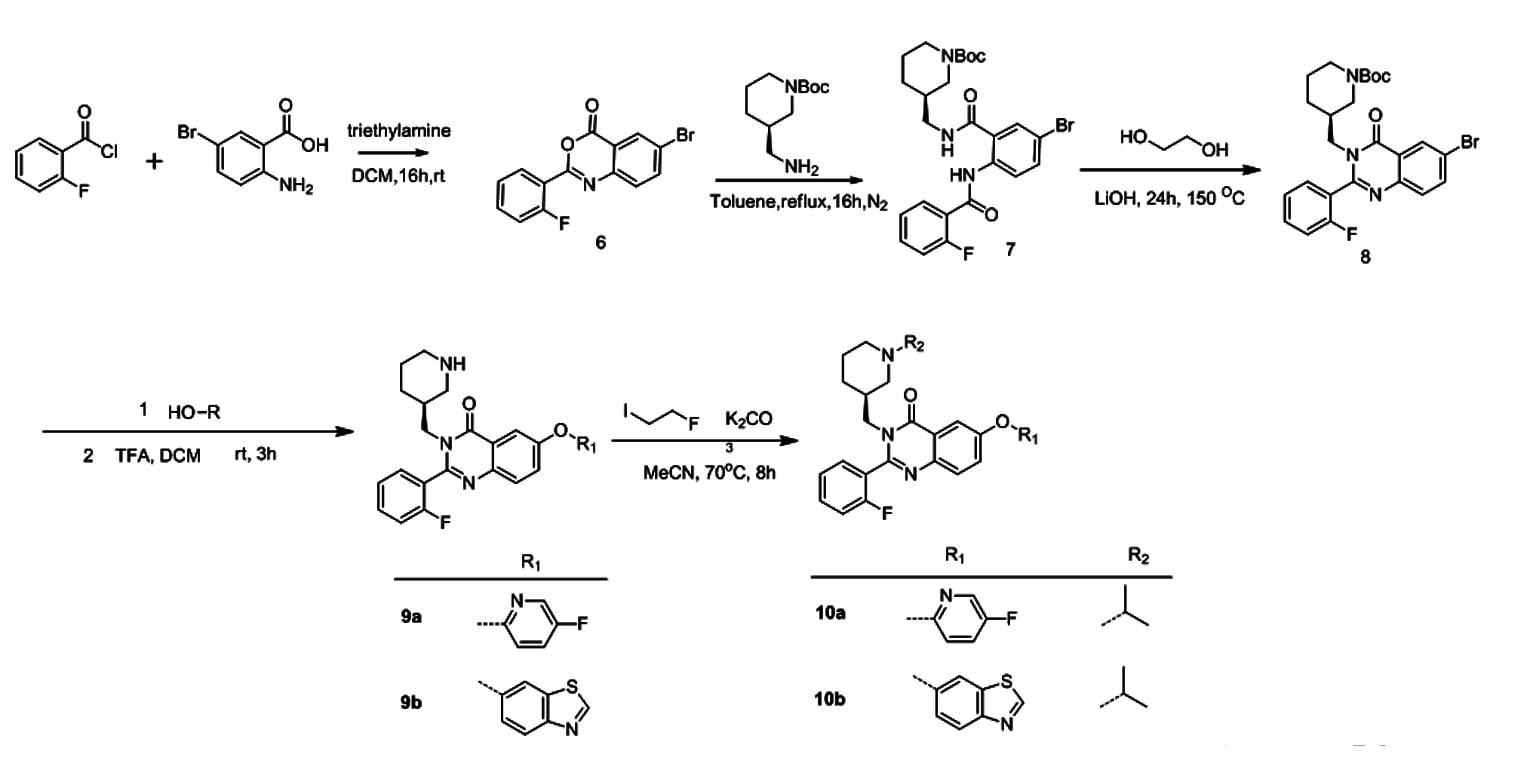
'Cold' Organic ChemistryWe perform muti-step organic synthesis to prepare molecules that we designed.
|
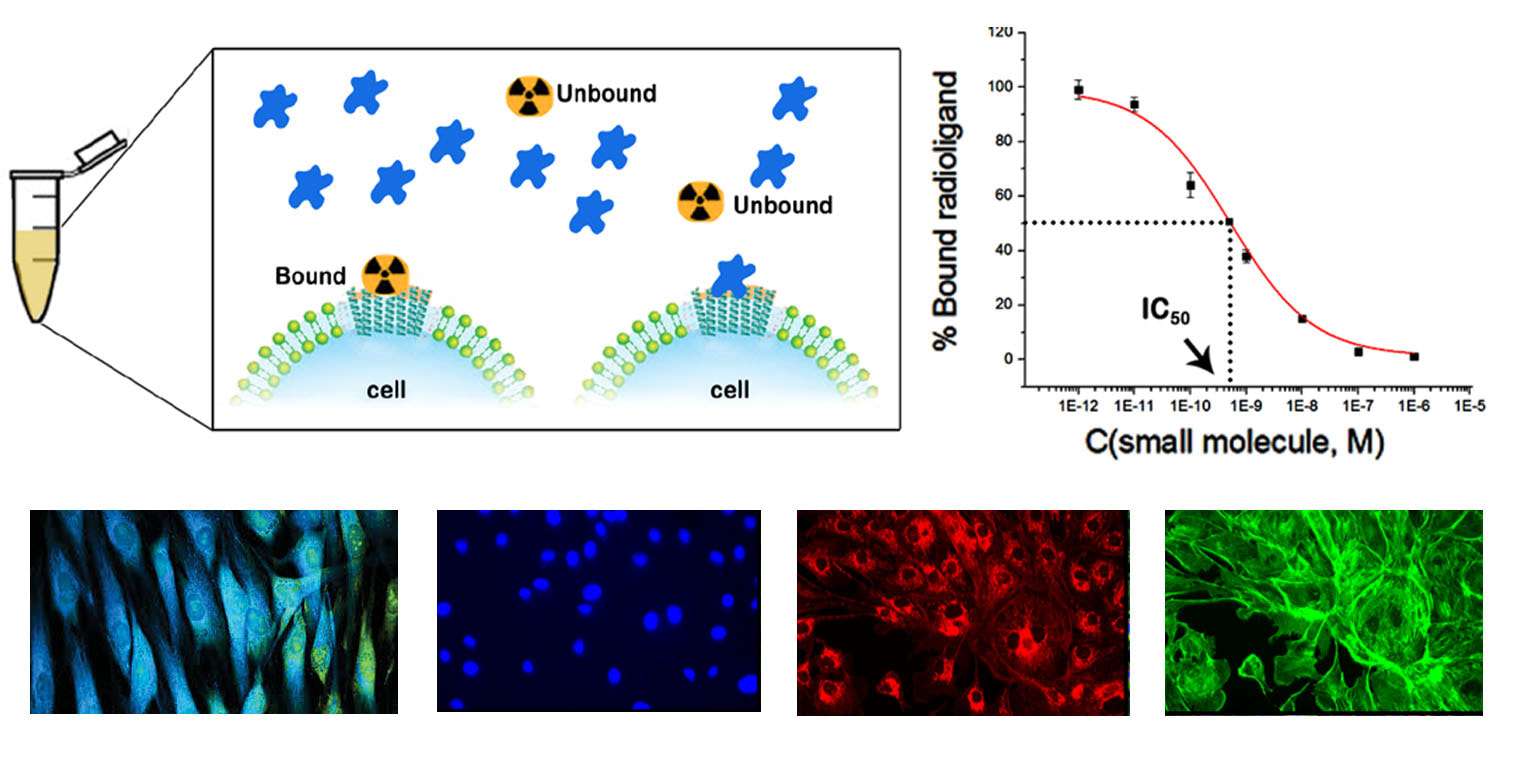
Biological evaluation in vitroCell culture and cell-based assay will be performed to evaluate the binding affinity and specificity of the synthesized molecules for their protein target. The candidate molecules with desire binding affinity will be selected for radiolabelling.
|
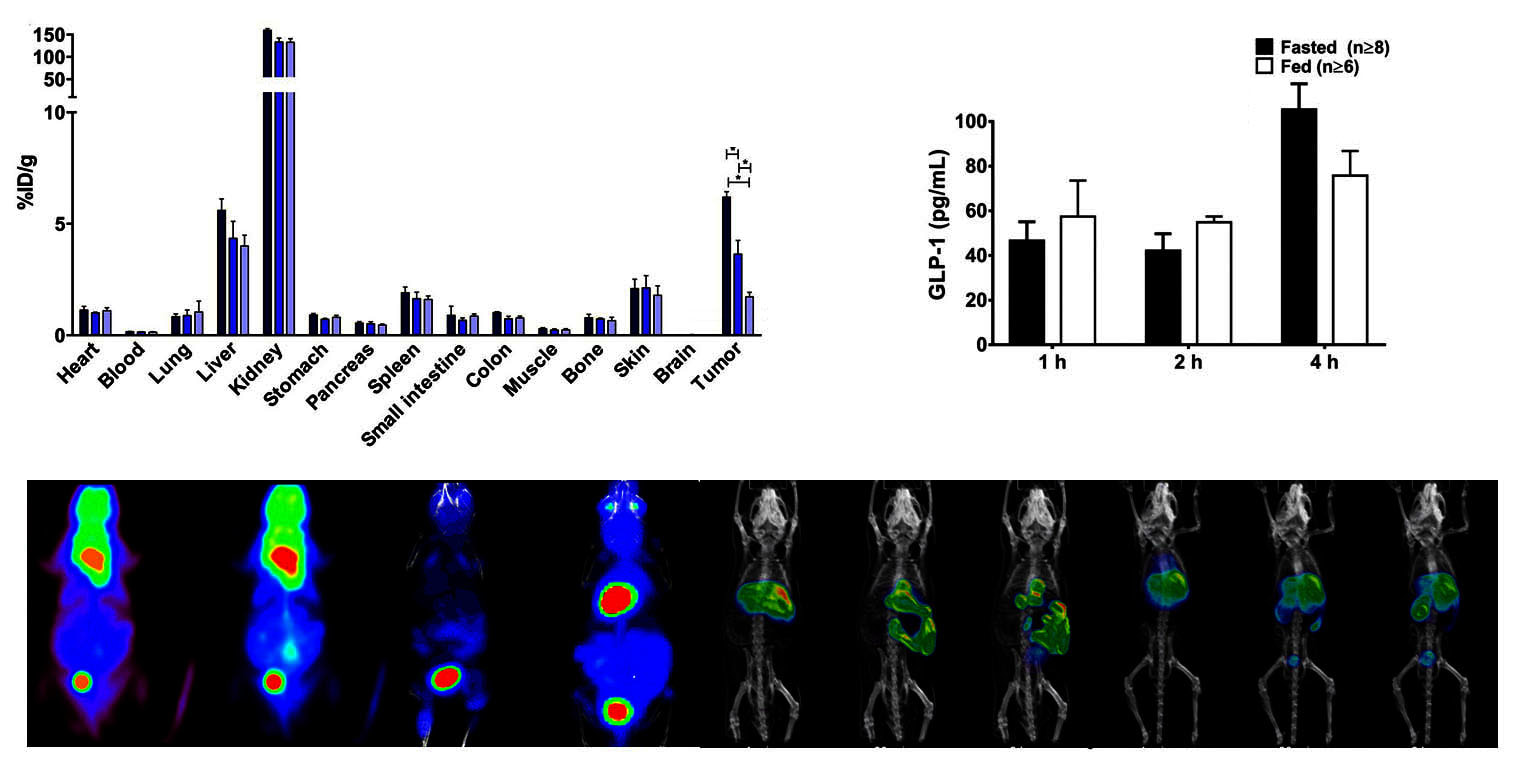
Animal studyPET imaging studies will be performed using mouse model of human disease to evaluate the radiolabelled molecules in vivo
|